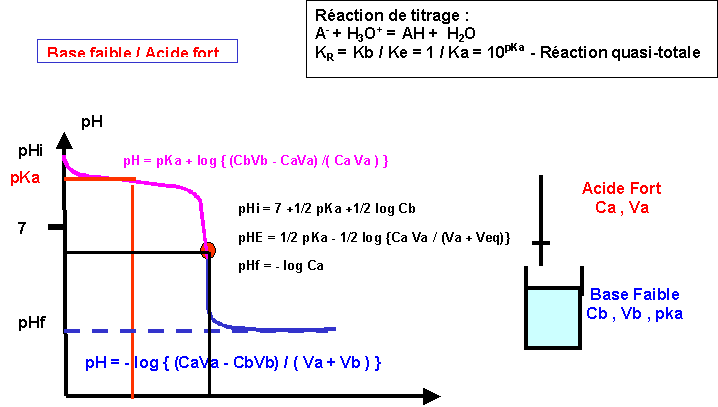
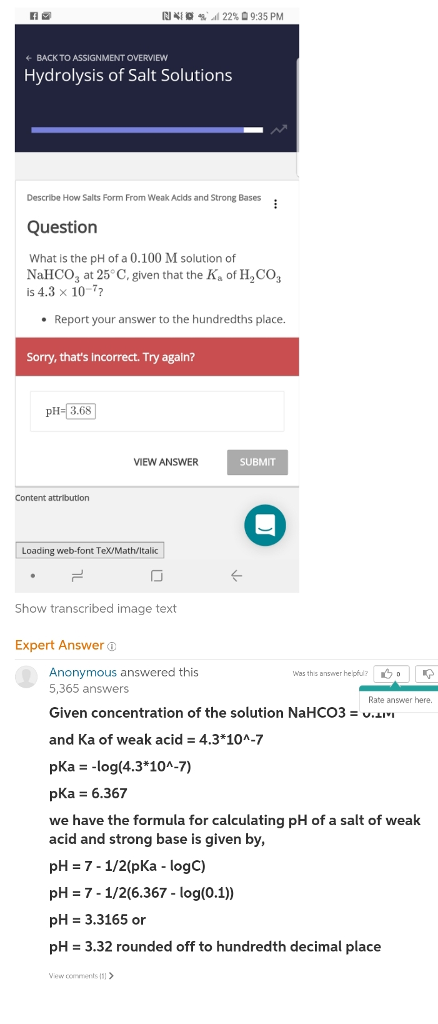
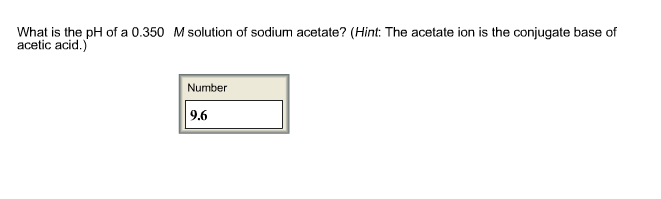
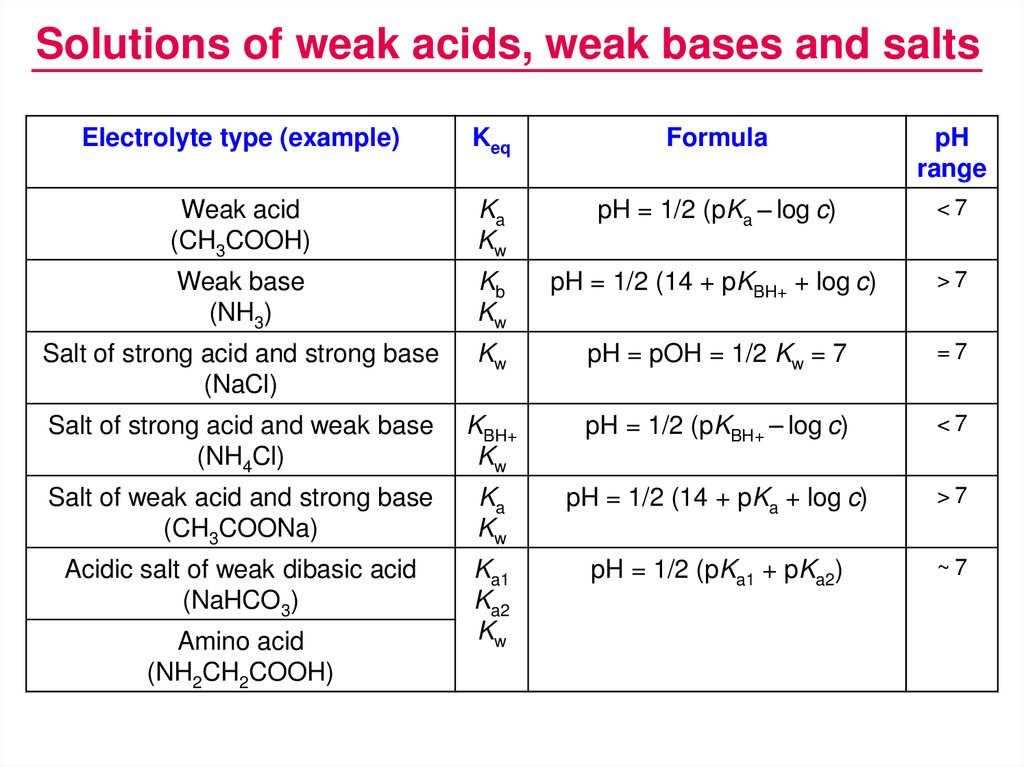
3) (NH),SO 40. pH of a salt of a strong base with weak acid (1) pH = K + pK, + logo (2) pH = LoK + Jok. 10g (3) pH = pkw + pka ---logc (4) None of these
![How is pH = 1/2[pKa - logc] - Chemistry - Chemical and Ionic Equilibrium - 13113047 | Meritnation.com How is pH = 1/2[pKa - logc] - Chemistry - Chemical and Ionic Equilibrium - 13113047 | Meritnation.com](https://s3mn.mnimgs.com/img/shared/ck-files/ck_57fe3e2aeb864.png)
How is pH = 1/2[pKa - logc] - Chemistry - Chemical and Ionic Equilibrium - 13113047 | Meritnation.com